EMH Schweizerischer Ärzteverlag AG
Farnsburgerstrasse 8
CH-4132 Muttenz
+41 (0)61 467 85 44
support@swisshealthweb.ch
www.swisshealthweb.ch
Publié le 06.10.2020
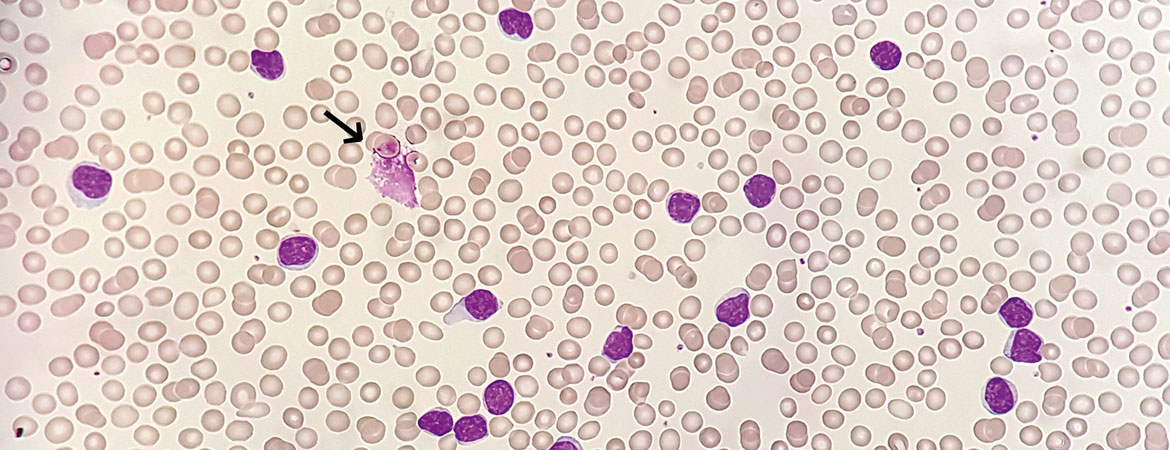
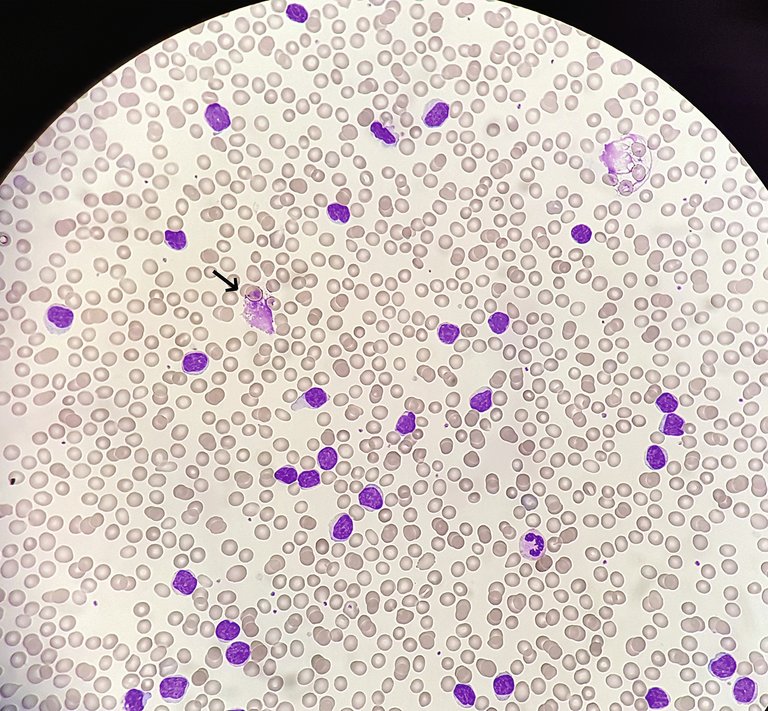
Aufgrund der Entwicklung neuer und sehr effektiver zielgerichteter Therapien bieten sich zunehmend mehr Behandlungsoptionen, die eine individualisierte Entscheidung vor Therapiebeginn einer CLL ermöglichen.
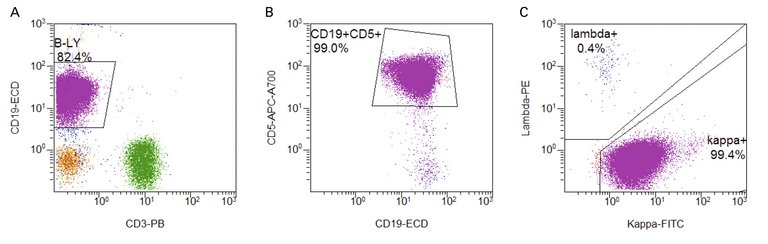
| Tabelle 1: Therapieindikation bei einer chronischen lymphatischen Leukämie (CLL)[7]. |
| Zunehmende Knochenmarkinsuffizienz mit progredienter Thrombozytopenie (Tc <100 G/l) oder Anämie (Hb <100 g/l) |
| Ausgeprägte oder zunehmende und symptomatische Splenomegalie |
| Massiv zunehmende oder symptomatische Lymphadenopathie |
| Zunehmende Lymphozytose mit 50%iger Zunahme innerhalb von 2 Monaten oder einer Verdopplungszeit von weniger als 6 Monaten unter Berücksichtigung der absoluten Lymphozytenzahl (relative Indikation) ausgehend von einem Basiswert von mindestens30 G/l Lymphozyten |
| Paraneoplastische Syndrome wiedie Autoimmunhämolytische Anämie(AIHA) oder Immunthrombozytopenie(ITP), dieschlecht auf Kortikosteroide ansprechen |
| Signifikante, durch die Krankheit verursachte Symptome («B-Symptome»: Nachtschweiss, Fieber, Fatigue, Gewichtsverlust) |
| Tabelle 2: Neue Therapieansätze. | |
| Neu zugelassene CD20-Antikörper | Obinutuzumab: in Kombination mit Chlorambucil höhere CR-Raten verglichen mit Rituximab (20,7 vs. 7,0%) sowie verbessertes OS und PFS [9]. |
| Ofatumumab: Ansprechraten bis zu 53% als Monotherapie [20]. | |
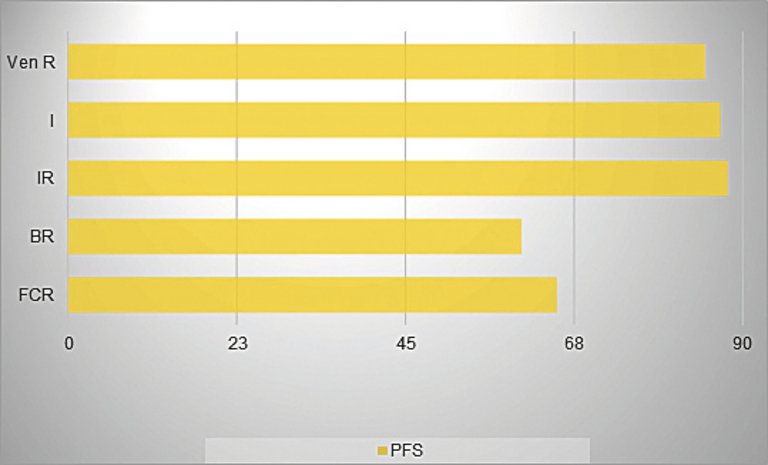
| BTK-Inhibitoren | Ibrutinib: in Kombination mit Rituximab besseres PFS und OS verglichen mit FCR, signifikant besseres PFS verglichen mit BR [11]. |
| Acalabrutinib (2.-Generations-BTK, noch keine Zulassung für CLL) Ansprechraten von 76% [21]. | |
| PI3-K-Inhibitoren | Idelalisib: Remissionsraten von bis zu 81% in Kombination mit Rituximab [22]. |
| Duvelisib: Ansprechraten von 74% [17]. | |
| Bcl-2-Inhibitor | Venetoclax: signifikant besseres PFS von Venetoclax/Rituximab (2-Jahres-PFS 84,9%) im Vergleich mit BR [15]. |
| CAR-T cells | Bislang erfolgreich eingesetzt bei refraktären Erkrankungen. Jedoch kleine Fallzahlen. Ansprechraten bis zu 70% [23]. |
| Lenalidomid | Immunmodulatorische Substanz, die jedoch bislang lediglich bei refraktären Erkrankungen eingesetzt wird. Keine randomisierten Studien verfügbar. Ansprechraten 32–47% [24, 25]. |
| BTK: Bruton-Tyrosinkinase; PI3-K: Phosphoinositid-3-Kinase; Bcl-2: B-cell lymphoma 2; CAR: chimeric antigen receptor; CR: complete response (vollständiges Ansprechen); OS: overall survival (Gesamtüberleben); PFS: progression-free survival (progressionsfreies Überleben); FCR: Fludarabin, Cyclophosphamid, Rituximab; BR: Bendamustin, Rituximab; CLL: chronische lymphatische Leukämie. | |
Publié sous la licence du droit d'auteur.
"Attribution - Non-Commercial - NoDerivatives 4.0"
Pas de réutilisation commerciale sans autorisation..
See: emh.ch/en/emh/rights-and-licences/