2 Hamre D, Procknow JJ. A new virus isolated from the human respiratory tract. Proc Soc Exp Biol Med. 1966;121(1):190–193.
doi:10.3181/00379727-121-307343 McIntosh K, Becker WB, Chanock RM. Growth in suckling-mouse brain of «IBV-like» viruses from patients with upper respiratory tract disease. Proc Natl Acad Sci U S A. 1967;58(6):2268–73.
doi:10.1073/pnas.58.6.22684 van der Hoek L, Pyrc K, Jebbink MF, Vermeulen-Oost W, Berkhout RJ, Wolthers KC, et al. Identification of a new human coronavirus. Nat Med. 2004;10(4):368–73.
doi: 10.1038/nm1024.
5 Woo PC, Lau SK, Chu CM, Chan KH, Tsoi HW, Huang Y, et al. Characterization and complete genome sequence of a novel coronavirus, coronavirus HKU1, from patients with pneumonia. J Virol. 2005;79(2):884–95.
doi:10.1128/JVI.79.2.884-895.2005.
6 Heikkinen T, Järvinen A. The common cold. Lancet. 2003;361(9351):51–9.
doi: 10.1016/S0140-6736(03)12162-9.
7 World Health Organization. Summary of probable SARS cases with onset of illness from 1 November 2002 to 31 July 2003 (based on data as of the 31 December 2003). (aufgerufen am 1.6.2020 von:
http://www.who.int/csr/sars/country/table2004_04_21/en/index.html)
8 Majumder MS, Rivers C, Lofgren E, Fisman D. Estimation of MERS-Coronavirus Reproductive Number and Case Fatality Rate for the Spring 2014 Saudi Arabia Outbreak: Insights from Publicly Available Data. PLoS Curr. 2014;6:ecurrents.outbreaks.98d2f8f3382d84f390736cd5f5fe133c. Published 2014 Dec 18.
doi:10.1371/currents.outbreaks.98d2f8f3382d84f390736cd5f5fe133c
9 Zhou P, Yang XL, Wang XG, Hu B, Zhang L, Zhang W, et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020;579(7798):270–73.
doi: 10.1038/s41586-020-2012-7.
10 Li C, Yang Y, Ren L. Genetic evolution analysis of 2019 novel coronavirus and coronavirus from other species [published online ahead of print, 2020 Mar 10]. Infect Genet Evol. 2020;82:104285.
doi:10.1016/j.meegid.2020.104285
11 Wu F, Zhao S, Yu B, Chen YM, Wang W, Song ZG, et al. A new coronavirus associated with human respiratory disease in China. Nature. 2020;579(7798):265–9.
doi: 10.1038/s41586-020-2008-3. Epub 2020 Feb 3. Erratum in: Nature. 2020;580(7803):E7.
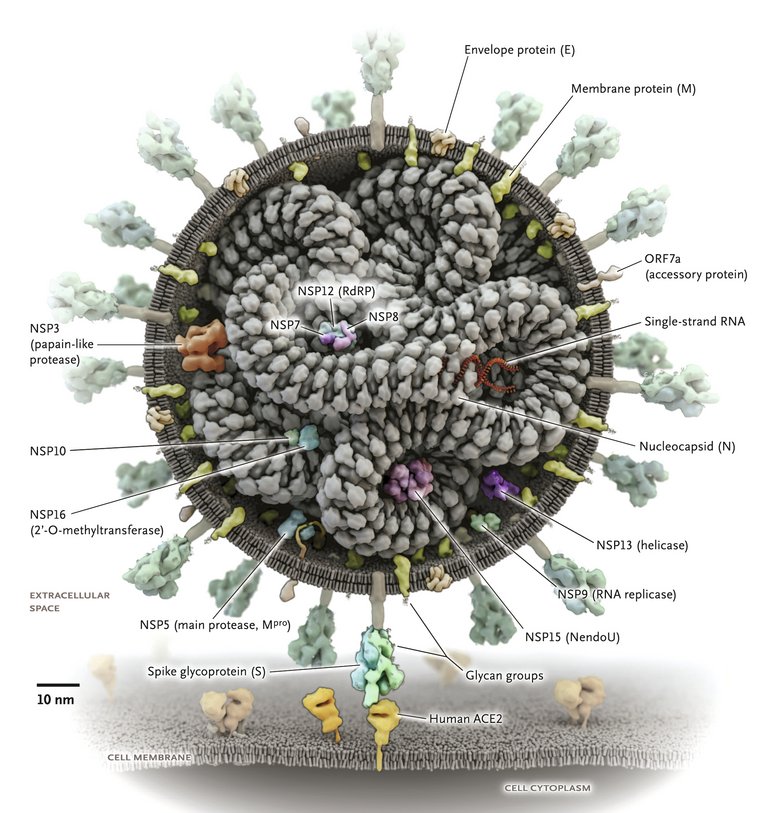
12 Yao H, Song Y, Chen Y, Wu N, Xu J, Sun C, et al. Molecular Architecture of the SARS-CoV-2 Virus. Cell. 2020 Oct 29;183(3):730–8.e13.
doi:10.1016/j.cell.2020.09.018. Epub 2020 Sep 6.
13 Ferretti L, Wymant C, Kendall M, Zhao L, Nurtay A, Abeler-Dörner L, et al. Quantifying SARS-CoV-2 transmission suggests epidemic control with digital contact tracing. Science. 2020 May 8;368(6491):eabb6936.
doi: 10.1126/science.abb6936. Epub 2020 Mar 31
14 Prather KA, Wang CC, Schooley RT. Reducing transmission of SARS-CoV-2. Science. 2020 May 27:eabc6197.
doi:10.1126/science.abc6197. Epub ahead of print.
15 Sungnak W, Huang N, Bécavin C, Berg M, Queen R, Litvinukova M, et al. SARS-CoV-2 entry factors are highly expressed in nasal epithelial cells together with innate immune genes. Nat Med. 2020;26(5):681–7.
doi:10.1038/s41591-020-0868-6. Epub 2020 Apr 23.
16 Hoffmann M, Kleine-Weber H, Schroeder S, Krüger N, Herrler T, Erichsen S, et al. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell. 2020;181(2):271–80.e8.
doi: 10.1016/j.cell.2020.02.052. Epub 2020 Mar 5.
17 Delang L, Abdelnabi R, Neyts J. Favipiravir as a potential countermeasure against neglected and emerging RNA viruses. Antiviral Res. 2018;153:85–94.
doi: 10.1016/j.antiviral.2018.03.003. Epub 2018 Mar 7.
18 Vincent MJ, Bergeron E, Benjannet S, Erickson BR, Rollin PE, Ksiazek TG, et al. Chloroquine is a potent inhibitor of SARS coronavirus infection and spread. Virol J. 2005 ;2:69.
doi: 10.1186/1743-422X-2-69.
19 Wang M, Cao R, Zhang L, Yang X, Liu J, Xu M, et al. Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro. Cell Res. 2020;30(3):269–71.
doi: 10.1038/s41422-020-0282-0. Epub 2020 Feb 4.
20 Yao X, Ye F, Zhang M, Cui C, Huang B, Niu P, et al. In Vitro Antiviral Activity and Projection of Optimized Dosing Design of Hydroxychloroquine for the Treatment of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2). Clin Infect Dis. 2020 Mar 9:ciaa237.
doi: 10.1093/cid/ciaa237. Epub ahead of print.
21 Gao J, Tian Z, Yang X. Breakthrough: Chloroquine phosphate has shown apparent efficacy in treatment of COVID-19 associated pneumonia in clinical studies. Biosci Trends. 2020;14(1):72–3.
doi:10.5582/bst.2020.01047. Epub 2020 Feb 19.
22 Liu M, Caputi TL, Dredze M, Kesselheim AS, Ayers JW. Internet Searches for Unproven COVID-19 Therapies in the United States. JAMA Intern Med. 2020 Apr 29:e201764.
doi: 10.1001/jamainternmed.2020.1764. Epub ahead of print.
24 Fiolet T, Guihur A, Rebeaud M, Mulot M, Peiffer-Smadja N, Mahamat-SalehY. Effect of hydroxychloroquine with or without azithromycin on the mortality of COVID-19 patients: a systematic review and meta-analysis. Clin Microbiol Infect. 2020 Aug 26;S1198-743X(20)30505-X.
https://doi.org/10.1016/j.cmi.2020.08.022.
25 Boulware DR, Pullen MF, Bangdiwala AS, Pastick KA, Lofgren SM, Okafor EC, et al. A Randomized Trial of Hydroxychloroquine as Postexposure Prophylaxis for Covid-19. N Engl J Med. 2020;383(6):517–25.
doi:10.1056/NEJMoa2016638. Epub 2020 Jun 3.
26 Cao B, Wang Y, Wen D, Liu W, Wang J, Fan G, et al. A Trial of Lopinavir-Ritonavir in Adults Hospitalized with Severe Covid-19. N Engl J Med. 2020;382(19):1787–99.
doi:10.1056/NEJMoa2001282. Epub 2020 Mar 18.
27 Chu CM, Cheng VC, Hung IF, Wong MM, Chan KH, Chan KS, et al. Role of lopinavir/ritonavir in the treatment of SARS: initial virological and clinical findings. Thorax. 2004;59(3):252–6.
doi:10.1136/thorax.2003.012658.
28 Hung IF, Lung KC, Tso EY, Liu R, Chung TW, Chu MY, et al. Triple combination of interferon beta-1b, lopinavir-ritonavir, and ribavirin in the treatment of patients admitted to hospital with COVID-19: an open-label, randomised, phase 2 trial. Lancet. 2020:S0140-6736(20)31042-4.
doi:10.1016/S0140-6736(20)31042-4. Epub ahead of print.
29 Warren TK, Jordan R, Lo MK, Ray AS, Mackman RL, Soloveva V, et al. Therapeutic efficacy of the small molecule GS-5734 against Ebola virus in rhesus monkeys. Nature. 2016;531(7594):381–5.
doi: 10.1038/nature17180. Epub 2016 Mar 2. Erratum in: ACS Chem Biol. 2016;11(5):1463.
30 Sheahan TP, Sims AC, Graham RL, Menachery VD, Gralinski LE, Case JB, et al. Broad-spectrum antiviral GS-5734 inhibits both epidemic and zoonotic coronaviruses. Sci Transl Med. 2017;9(396):eaal3653.
doi:10.1126/scitranslmed.aal3653.
31 Mulangu S, Dodd LE, Davey RT Jr, Tshiani Mbaya O, Proschan M, Mukadi D, et al. A Randomized, Controlled Trial of Ebola Virus Disease Therapeutics. N Engl J Med. 2019;381(24):2293–303.
doi:10.1056/NEJMoa1910993. Epub 2019 Nov 27.
32 Beigel JH, Tomashek KM, Dodd LE, Mehta AK, Zingman BS, Kalil AC, et al. Remdesivir for the Treatment of Covid-19 - Preliminary Report. N Engl J Med. 2020 May 22.
doi: 10.1056/NEJMoa2007764. Epub ahead of print.
33 Wang Y, Zhang D, Du G, Du R, Zhao J, Jin Y, et al. Remdesivir in adults with severe COVID-19: a randomised, double-blind, placebo-controlled, multicentre trial. Lancet. 2020;395(10236):1569–78.
doi:10.1016/S0140-6736(20)31022-9. Epub 2020 Apr 29.
34 A Trial of Remdesivir in Adults With Mild and Moderate COVID-19. NCT04252664. (aufgerufen am 1.6.2020 von
www.clinicaltrials.gov
35 Adaptive COVID-19 Treatment Trial 2 (ACTT-II). NCT04401579. (aufgerufen am 1.6.2020 von
www.clinicaltrials.gov)
36 Study of Merimepodib in Combination With Remdesivir in Adult Patients With Advanced COVID-19. NCT04410354. (aufgerufen am 1.6.2020 von
www.clinicaltrials.gov)
37 A Study to Evaluate the Efficacy and Safety of Remdesivir Plus Tocilizumab Compared With Remdesivir Plus Placebo in Hospitalized Participants With Severe COVID-19 Pneumonia (REMDACTA). NCT04409262. (aufgerufen am 1.6.2020 von
www.clinicaltrials.gov)
40 McKee DL, Sternberg A, Stange U, Laufer S, Naujokat C. Candidate drugs against SARS-CoV-2 and COVID-19. Pharmacol Res. 2020;157:104859.
doi:10.1016/j.phrs.2020.104859. Epub ahead of print.
41 Kawase M, Shirato K, van der Hoek L, Taguchi F, Matsuyama S. Simultaneous treatment of human bronchial epithelial cells with serine and cysteine protease inhibitors prevents severe acute respiratory syndrome coronavirus entry. J Virol. 2012;86(12):6537–45.
doi:10.1128/JVI.00094-12. Epub 2012 Apr 11.
42 Talukdar R, Tandon RK. Pancreatic stellate cells: new target in the treatment of chronic pancreatitis. J Gastroenterol Hepatol. 2008;23(1):34–41.
doi: 10.1111/j.1440-1746.2007.05206.x. Epub 2007 Nov 6.
43 Chen G, Wu D, Guo W, Cao Y, Huang D, Wang H, et al. Clinical and immunological features of severe and moderate coronavirus disease 2019. J Clin Invest. 2020;130(5):2620–9.
doi:10.1172/JCI137244.
44 Channappanavar R, Perlman S. Pathogenic human coronavirus infections: causes and consequences of cytokine storm and immunopathology. Semin Immunopathol. 2017;39(5):529–39.
doi:10.1007/s00281-017-0629-x. Epub 2017 May 2.
45 Krüttgen A, Rose-John S. Interleukin-6 in sepsis and capillary leakage syndrome. J Interferon Cytokine Res. 2012;32(2):60–5.
doi:10.1089/jir.2011.0062. Epub 2011 Dec 2.
46 Ruan Q, Yang K, Wang W, Jiang L, Song J. Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan, China. Intensive Care Med. 2020;46(5):846–8.
doi: 10.1007/s00134-020-05991-x. Epub 2020 Mar 3. Erratum in: Intensive Care Med. 2020 Apr 6.
47 Zhou F, Yu T, Du R, Fan G, Liu Y, Liu Z, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395(10229):1054–62.
doi:10.1016/S0140-6736(20)30566-3. Epub 2020 Mar 11. Erratum in: Lancet. 2020 Mar 28;395(10229):1038. Erratum in: Lancet. 2020 Mar 28;395(10229):1038.
48 Dagens A, Sigfrid L, Cai E, Lipworth S, Cheng V, Harris E, et al. Scope, quality, and inclusivity of clinical guidelines produced early in the covid-19 pandemic: rapid review. BMJ. 2020;369:m1936.
doi:10.1136/bmj.m1936. Erratum in: BMJ. 2020 Jun 12;369:m2371.
49 RECOVERY Collaborative Group, Horby P, Lim WS, Emberson JR, Mafham M, Bell JL, Linsell L, et al. Dexamethasone in Hospitalized Patients with Covid-19 – Preliminary Report. N Engl J Med. 2020 Jul 17:NEJMoa2021436.
doi: 10.1056/NEJMoa2021436. Epub ahead of print.
51 Xu XL, Han MF, Li TT, Sun W, Wang DS, Fu BQ, et al. Effective treatment of severe COVID-19 patients with tocilizumab. ChinaXiv. 2020;202003.00026 (aufgerufen am 1.6.2020 von
http://www.chinaxiv.org/abs/202003.00026)
52 Tocilizumab in the Treatment of Coronavirus Induced Disease (COVID-19). NCT04335071 (aufgerufen am 1.6.2020 von
www.clinicaltrials.gov)
53 von Behring E, Kitasato S. Ueber das Zustandekommen der Diphtherie-Immunität und der Tetanus-Immunität bei Thieren [The mechanism of diphtheria immunity and tetanus immunity in animals. 1890]. Mol Immunol. 1991;28(12):1317, 1319–20. German.
54 Hey A. History and Practice: Antibodies in Infectious Diseases. Microbiol Spectr. 2015;3(2):AID-0026-2014.
doi: 10.1128/microbiolspec.AID-0026-2014.
55 Soo YO, Cheng Y, Wong R, Hui DS, Lee CK, Tsang KK, et al. Retrospective comparison of convalescent plasma with continuing high-dose methylprednisolone treatment in SARS patients. Clin Microbiol Infect. 2004;10(7):676–8.
doi:10.1111/j.1469-0691.2004.00956.x.
56 WHO. Use of convalescent whole blood or plasma collected from patients recovered from Ebola virus disease for transfusion, as an empirical treatment during outbreaks. (aufgerufen am 1.6.2020 von
http://apps.who.int/iris/rest/bitstreams/604045/retrieve)
57 Arabi Y, Balkhy H, Hajeer AH, Bouchama A, Hayden FG, Al-Omari A, et al. Feasibility, safety, clinical, and laboratory effects of convalescent plasma therapy for patients with Middle East respiratory syndrome coronavirus infection: a study protocol. Springerplus. 2015 Nov 19;4:709.
doi:10.1186/s40064-015-1490-9.
58 Hung IF, To KK, Lee CK, Lee KL, Chan K, Yan WW, et al. Convalescent plasma treatment reduced mortality in patients with severe pandemic influenza A (H1N1) 2009 virus infection. Clin Infect Dis. 2011;52(4):447–56.
doi:10.1093/cid/ciq106. Epub 2011 Jan 19.
59 Li L, Zhang W, Hu Y, Tong X, Zheng S, Yang J, et al. Effect of Convalescent Plasma Therapy on Time to Clinical Improvement in Patients With Severe and Life-threatening COVID-19: A Randomized Clinical Trial. JAMA. 2020;324(5):1–11.
doi: 10.1001/jama.2020.10044. Epub ahead of print. Erratum in: JAMA. 2020 Aug 4;324(5):519.